
Overview
The pharmaceutical industry faces unique challenges during the winter months, primarily due to the harsh environmental conditions that can significantly impact the stability and integrity of pharmaceutical products. As temperatures drop and fluctuate, and as heating systems struggle to maintain a consistent environment, the risk to temperature-sensitive medications increases.
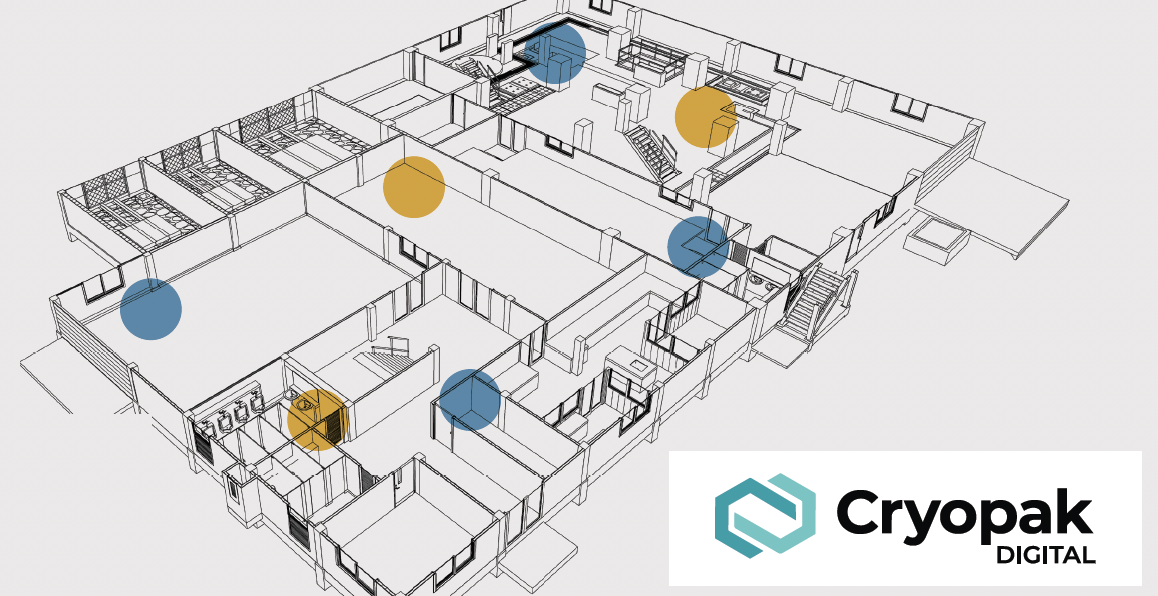
The Role of Temperature Mapping in Regulatory Compliance
Storage conditions for pharmaceutical products and materials should be in compliance with the product labelling, which is based on the results of stability testing storage conditions. All drugs should be stored according to the conditions described on the label. Pharmaceutical facilities are required to maintain specific environmental conditions for storing medications and vaccines. Maintaining a uniform temperature is not just a best practice; it’s a regulatory requirement, essential for compliance with Health Canada and the FDA guidelines. The recent changes in the FDA and Canadian regulations (GUI-0069 and various federal CFR codes), along with the objective to eliminate the waste of time and money to fix environmental conditions failures has made temperature mapping an integral aspect of any storage area operation. By Using iMini data loggers and our CFR 21 Part 11 compliant ConsolePlus software, Cryopak Digial meticulously monitor temperature and humidity, ensuring optimal conditions in diverse environments – be it a pharmaceutical facility, food storage and warehouse.


Read the Full White Paper Here: Cryopak Digital Winter Mapping White Paper

